A new European authorisation redefining mole repellent solutions
Discover why this new European approval could transform the mole repellent market through a simpler, safer, and scientifically validated approach.

The mole repellent market is undergoing a major shift with the recent acquisition, in March, of a new European authorisation by Armosa for PT19 biocidal products. This approval reflects our ongoing commitment to innovation, but above all our ambition to transform a category that has long been perceived as limited in terms of reliable and easy-to-use solutions.
A turning point for the market, backed by scientific evidence
Obtaining this European authorisation goes far beyond a simple administrative validation. It is the result of:
- Rigorous testing under real-life conditions,
- Scientific validation of efficacy,
- Demonstration of product safety.
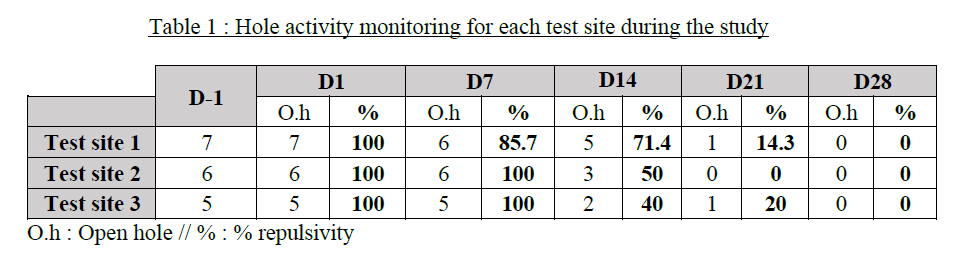
Field trials conducted across three distinct environments, targeting Talpa europaea moles, show a progressive reduction in activity. Effects are visible from the first few days, leading to a complete disappearance of activity signs after several weeks, reflecting a gradual behavioural change in the animals.
.png)
Why the market needed innovation?
From a field perspective, we have observed for several years that the mole control market faces several limitations:
- Trapping, while effective, is restrictive and not always acceptable for some users (animal killing, carcass handling, operational constraints).
- Alternative solutions, such as ultrasonic devices, are often more expensive and may represent a barrier for some users.
- A lack of solutions combining simplicity, safety and proven effectiveness.
It is precisely to address these challenges that we have developed a new approach, aligned with current market expectations.
An effective repellent solution, designed for the user
Our granule-based solution is formulated using plant-based active substances, including peppermint and lavender essential oils.
In practical terms, this means:
- Simple and fast application, with no specific constraints (using a 3 g dosing spoon directly in the tunnels).
- Proven efficacy for up to 7 days, validated for both professional and non-professional use.
- A safe, low-risk alternative for consumer (no hazard pictograms), non-toxic compared to conventional mole control methods.
- A behavioural repellent action, encouraging moles to leave treated tunnels.
From regulation to market: Armosa expertise supporting our partners
Beyond the product itself, we master the entire value chain: formulation, regulatory compliance, production, logistics and commercial support.
Armosa’s expertise enables us to offer:
- Approved solutions ready for distribution,
- Private label (OEM) development capabilities,
- Optimised time-to-market,
- Support tailored to regulatory and commercial challenges.
In a constantly evolving environment, we position ourselves as a strategic partner to help strengthen your product offering. This new European authorisation does not simply introduce a new product — it contributes to raising the standards of the market.
For our partners, this represents an opportunity to integrate innovative, scientifically validated solutions that are fully aligned with current expectations.
Want to learn more about our solutions and expertise? Contact our team!


